
Calculated by the following equation: where V is the number of valence electrons of an isolated. You have signed an examinee agreement, and it will be enforced on this subreddit.ĭo not intentionally advertise paid or free products or services of any sort. The formal charge of any atom in a molecule can be calculated. We have one "stickied" post for each exam and score release day, contain all test day discussion/reactions to that thread only.ĭo not discuss any specific information from your actual MCAT exam. For an example format for submitting pictures of questions from practice material click hereĭo not link to content that infringes on copyright laws (MCAT torrents, third party resources, etc).ĭo not post repeat "GOOD LUCK", "TEST SCORE", or test reaction posts. These are considered spoilers and should be marked as such. Be nice to each other, hating on other users won't help you get extra points on the MCAT, so why do it?ĭo not post any question information from any resource in the title of your post. Rudeness or trolling will not be tolerated. Here you will find a clear explanation and illustration of how to calculate formal charge of an atom, which is part of a molecule. The formal charge of an atom in a molecule is the hypothetical charge the atom would have if we could redistribute the electrons in the bonds evenly between the atoms. Please message the moderators with your skills/ideas! MCAT RESOURCES & INFO Study Groups If you are looking for guidelines on calculating formal charge of an atom in a molecule, this article has what you are looking for. We can use the concept of formal charges to help us predict the most appropriate Lewis structure when more than one is reasonable. The following equation can calculate the formal charge of an atom in a molecule.  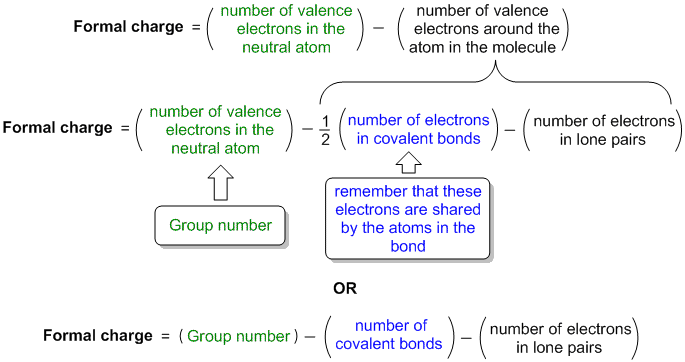 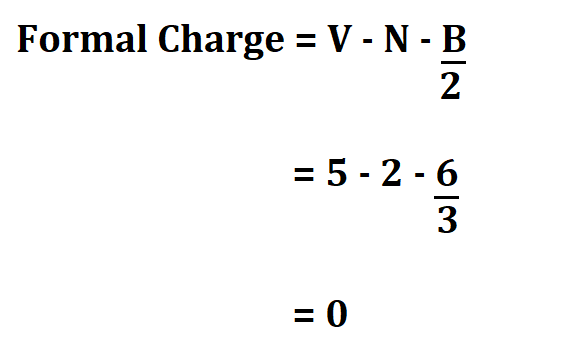
Want to help us improve this subreddit or tell us about a new resource we can add to the sidebar? The formula for calculating the formal charge on an atom is simple. The formula for calculating the formal charge on an atom is simple. 3 for boron, 4 for carbon, 5 for nitrogen, and so on). C Valence electrons Nonbonding electrons- Bonding electrons/2. evaluating the number of valence electrons (VE) the neutral atom has (e.g. an atom in a molecule 9.6.5 Using formal charge to determine molecular structure. To calculate the formal charge of an atom, we start by. Here is the formula: Formal Charge V N (B/2) In this formula, V stands for the number of valence electrons of that atom ( these are the electrons that revolve in the outermost orbit of the atom ), N stands for the number of non-bonded electrons, and B. Calculating formal charge for the resonance structures of N2O Lewis. Below you will find our forum rules, resources, and more. You can picture the chemical bond using a Lewis structure diagram of the compound. We request that you read the sidebar COMPLETELY before you post. r/MCAT is a place for support, discussion, advice, social networking, news, study tips and more. The MCAT (Medical College Admission Test) is offered by the AAMC and is a required exam for admission to medical schools in the USA and Canada. Fortunately, this only requires some practice with recognizing common bonding patterns.Welcome to the BEST place for MCAT prep and practice materials. You need to develop the ability to quickly and efficiently draw large structures and determine formal charges. As you get more experience with organic structures, you will be able to quickly look at this type of complicated structure and determine charges on each atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
